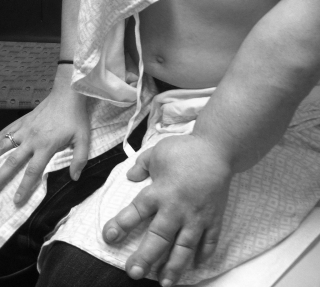
Researchers at the School of Medicine have developed a possible treatment for lymphedema, the severe swelling of an arm or leg that can occur when the lymph system is blocked. Using scaffolding composed of specially patterned collagen nanofibers, the researchers coaxed lymph vessels to grow around lymph blockages.
The technique was effective at treating lymphedema in pigs, the scientists report in a study published online June 7 in Biomaterials.
“We were able to take a cue from nature about what molecules spur vessel growth, but also think outside the box and use this nanoscale scaffolding to bridge the blockages,” said Ngan Huang, PhD, assistant professor of cardiothoracic surgery and a co-senior author of the study. “I think combining the two was really key.”